syjiaxuan's answer to Annela's Secondary 4 Chemistry Singapore question.
done
{{ upvoteCount }} Upvotes
clear
{{ downvoteCount * -1 }} Downvotes

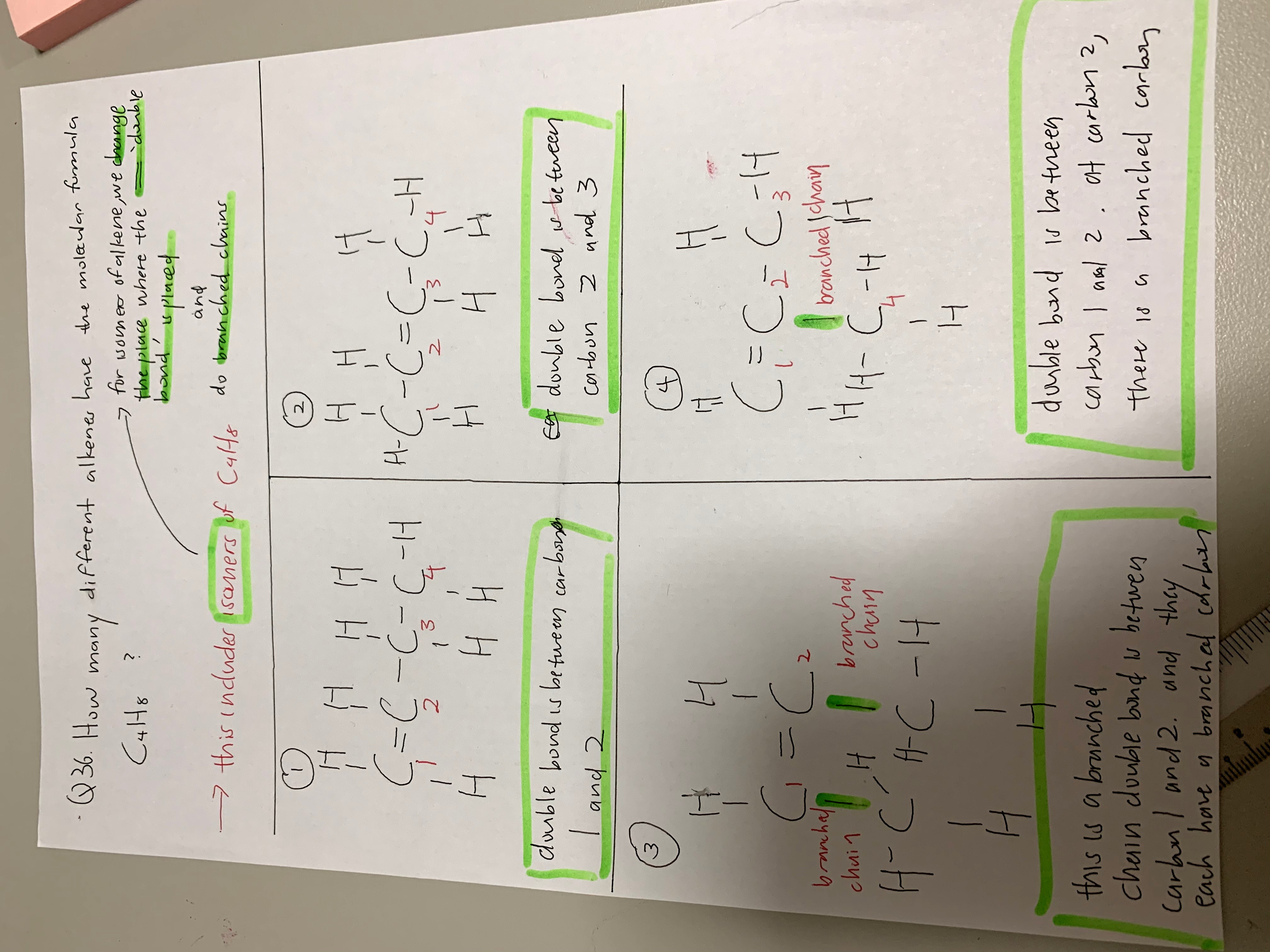
Hope this helps! Impt parts in green !
Date Posted:
4 years ago
Your third case listed is approximately the same as the second case if cis/trans configuration did not exist. Because the carbon atoms run continuously in a path (think of connecting the dots, where each C is a dot, without crossing over a drawn line or lifting your pencil).
Because the carbon at the “bottom” sides can be placed on the left side and right side without making much difference.
As an O Level student I would put 3 isomers. As an A level student I would put 4 isomers.
Because the carbon at the “bottom” sides can be placed on the left side and right side without making much difference.
As an O Level student I would put 3 isomers. As an A level student I would put 4 isomers.